How Joenja works
Joenja is designed to correct the underlying immune defect caused by APDS to help normalize the hyperactive PI3Kδ pathway1-3
HOW JOENJA WORKS
Joenja is the first and only precision therapy that corrects the underlying immune defect in patients 4 years and older with APDS1,2,4
Only Joenja specifically targets and blocks the PI3Kδ protein, addressing immune dysregulation and deficiency at the earliest stage of the PI3K pathway1-3
PI3Kδ signaling in B and T cells
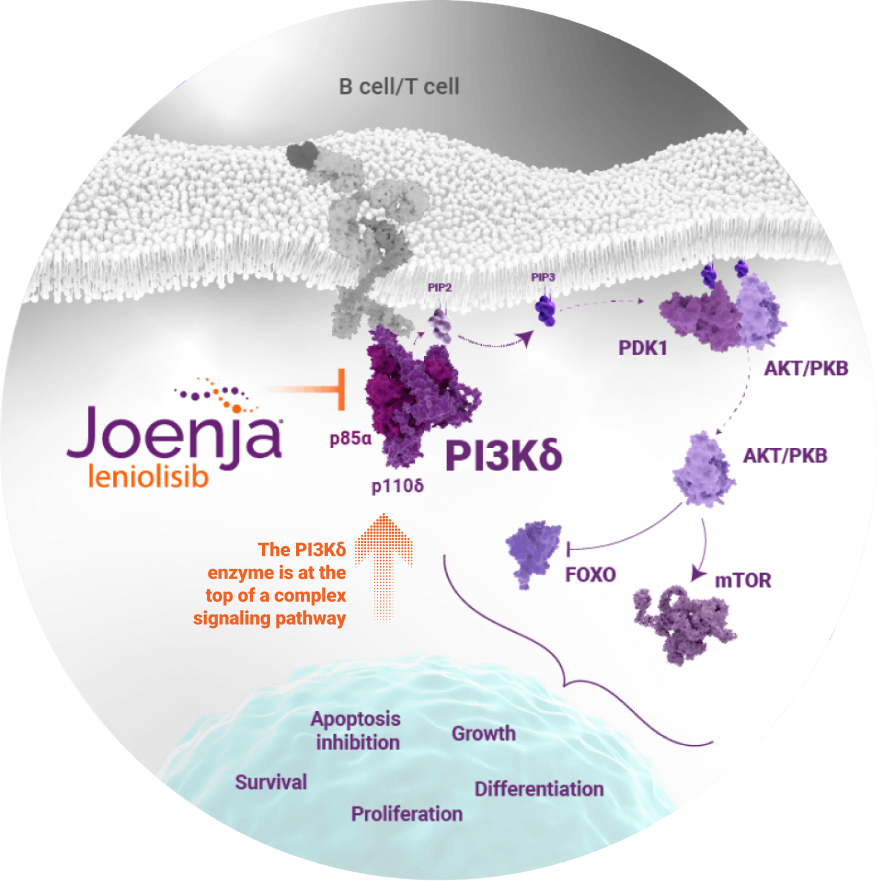
Note: Illustration does not include all steps in the signaling pathway.
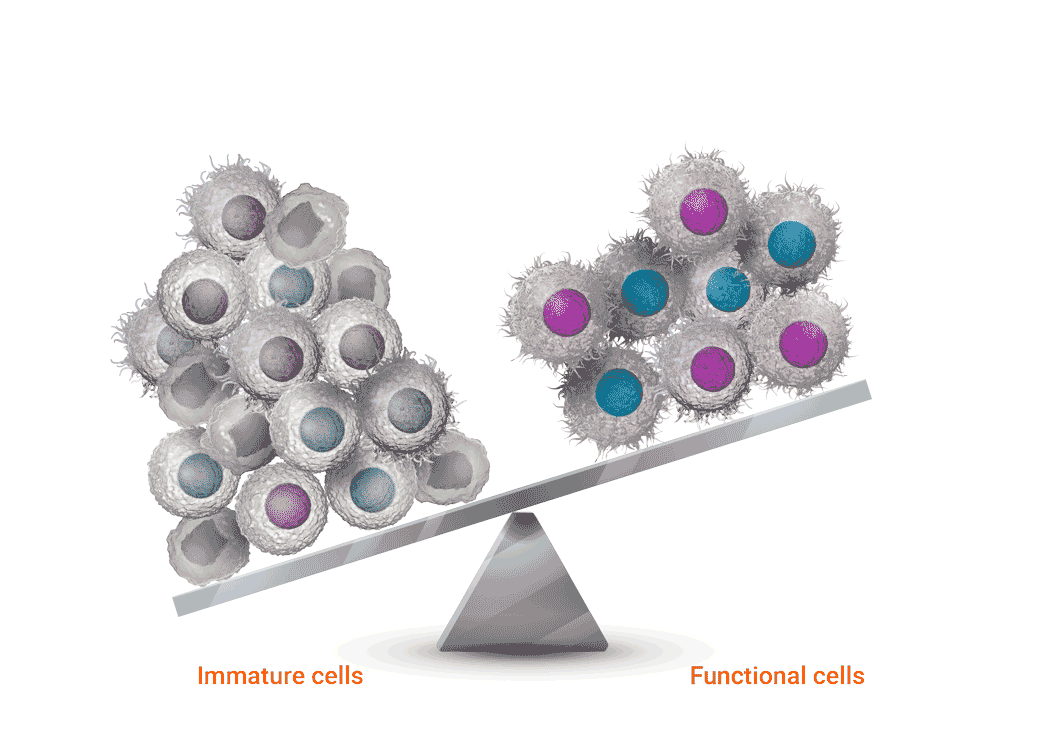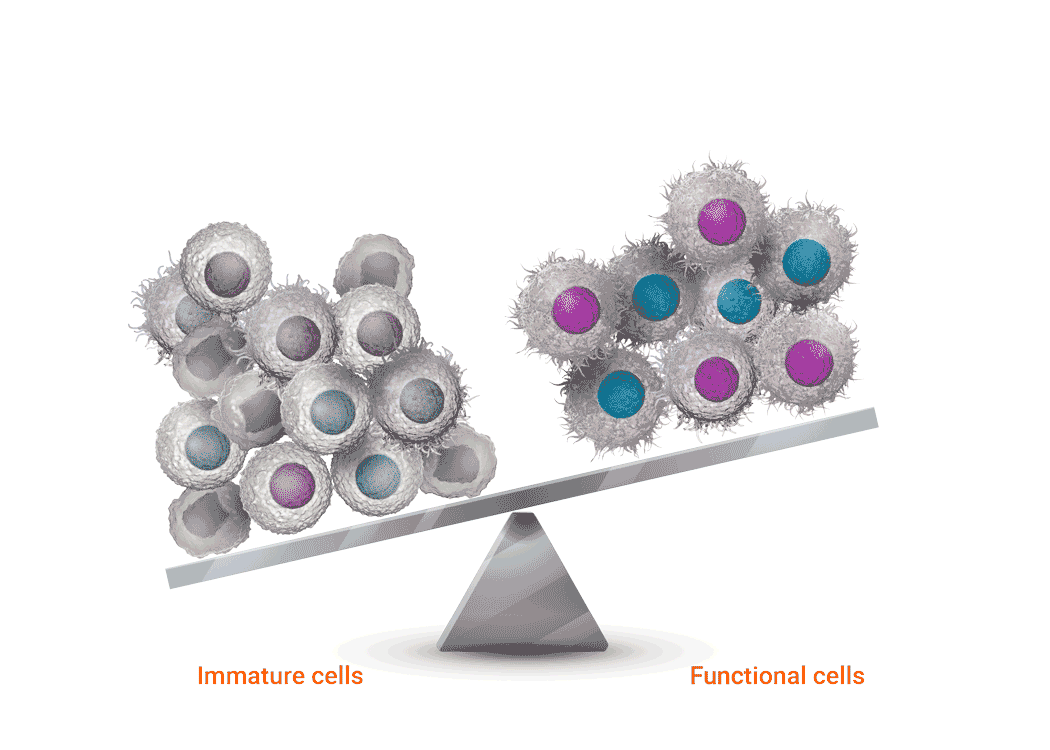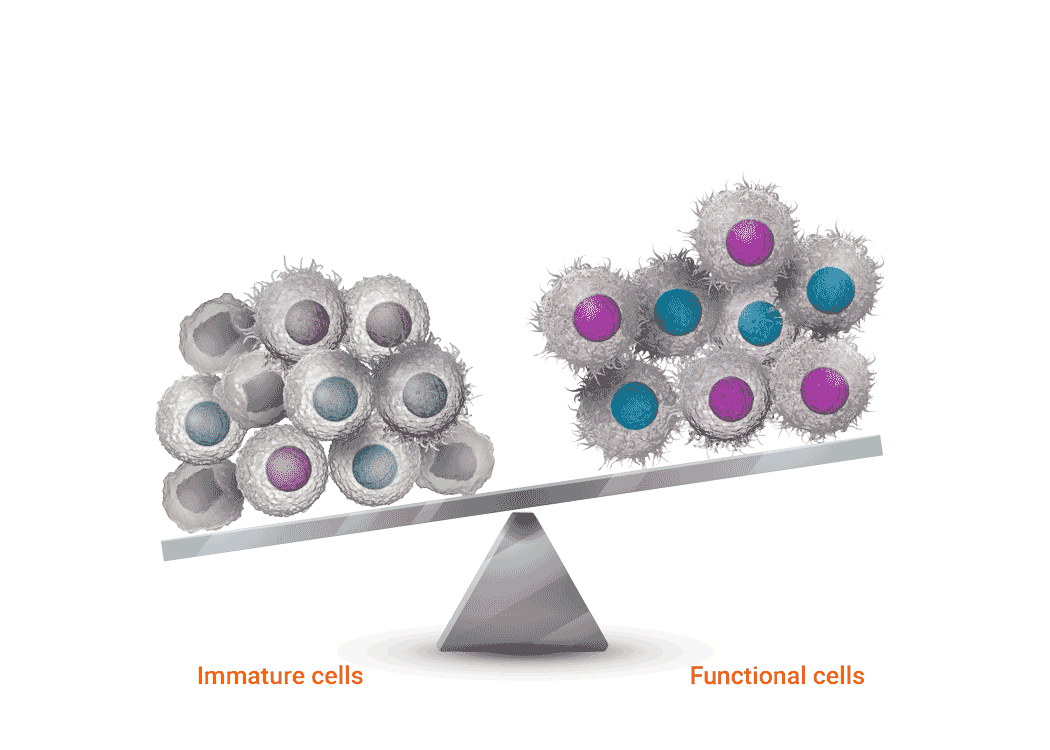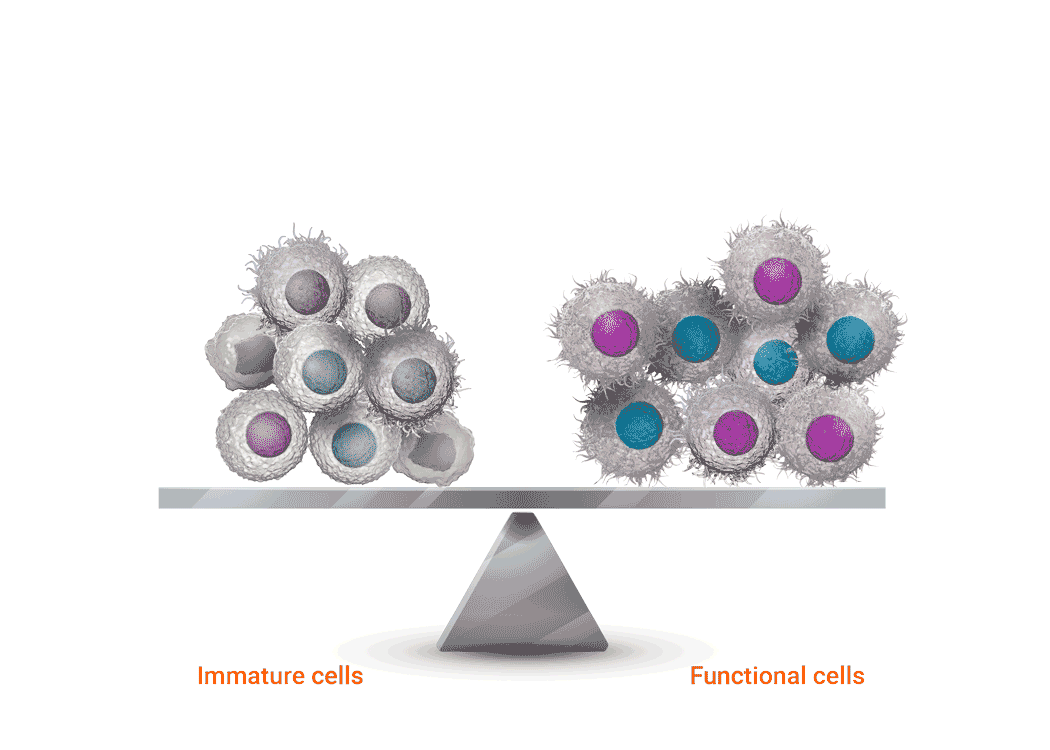
Joenja facilitates a balanced PI3Kδ pathway to support proper immune function1-3
JOENJA IN ACTION
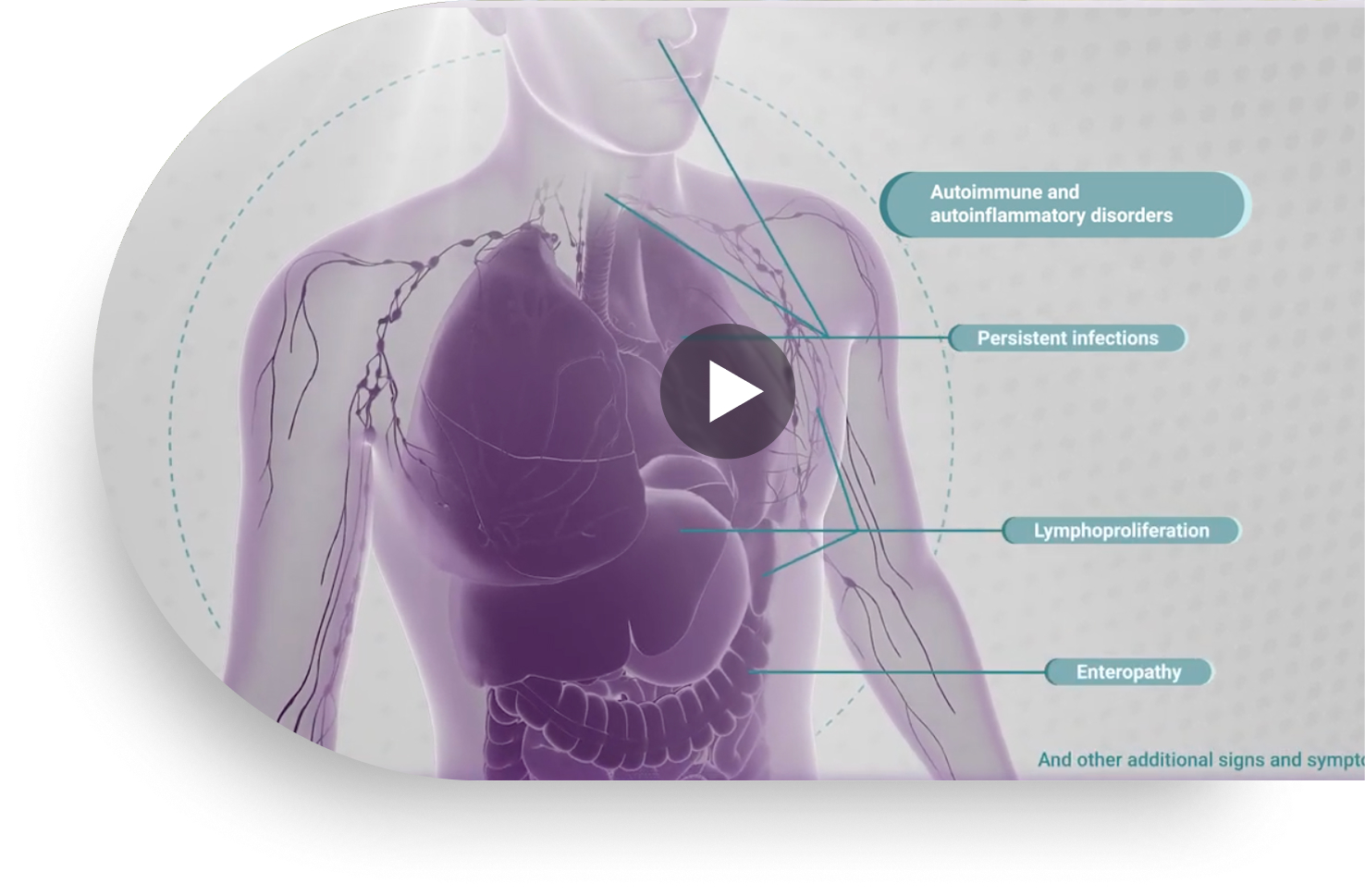
Joenja mechanism of action
This video provides a detailed illustration of the mechanism of action for Joenja and shows how only Joenja addresses both immune deficiency AND immune dysregulation to help normalize immune balance.
Efficacy Results (Adolescents & Adults 12+)
→
APDS Assist
→
AKT/PKB, protein kinase B; APDS, activated PI3Kδ syndrome; FOXO, forkhead box O; mTOR, mammalian target of rapamycin; p85α, the regulatory subunit of the PI3Kδ enzyme; p110δ, the catalytic subunit of the PI3Kδ enzyme; PDK1, phosphoinositide-dependent kinase 1; PIP2, phosphatidylinositol (4,5)-bisphosphate; PIP3, phosphatidylinositol (3,4,5)-trisphosphate.
References: 1. Joenja (leniolisib). Prescribing information. Pharming Healthcare, Inc; 2026. 2. Rao VK, Webster S, Šedivá A, et al. A randomized, placebo-controlled phase 3 trial of the PI3Kδ inhibitor leniolisib for activated PI3Kδ syndrome. Blood. 2023;141(9):971-983. doi:10.1182/blood.2022018546 3. Rao VK, Webster S, Šedivá A, et al. A randomized, placebo-controlled phase 3 trial of the PI3Kδ inhibitor leniolisib for activated PI3Kδ syndrome. Supplemental. Blood. 2023;141(9):971-983. doi:10.1182/blood.2022018546 4. Pharming Group. Pharming announces US FDA approval of Joenja® (leniolisib) as the first and only treatment indicated for APDS. Press release. March 24, 2023. Accessed January 9, 2026. https://www.pharming.com/news/pharming-announces-us-fda-approval-joenja-leniolisib-first-and-only-treatment-indicated-apds.