Joenja results
Adolescents & adults 12+
Joenja showed significant improvements for patients 12+ with APDS
CLINICAL TRIAL DESIGN
The Joenja clinical trials for people 12+ years of age1-4
Part 1: Dose Finding
Timing
12 weeks
Participants
6 patients
Age
Patients 12+
Conditions
- Non-randomized, open-label, dose-finding study in 6 patients with APDS; dose range was 10 mg, 30 mg, and 70 mg BID for 4 weeks at each dose
- Oral dose 70 mg BID selected for Part 2
Part 2: Efficacy and Safety Evaluation
Timing
Randomized period of 12 weeks
Participants
31 participants
Age
Patients 12+
Conditions
- Randomized, triple-blinded (patient, caregiver, investigator), placebo-controlled, fixed-dose study of 70 mg BID
- Co-primary efficacy end points (improvement in lymphoproliferation and normalization of immunophenotype)
- Change from baseline in the log10-transformed SPD of index lesions
- Change from baseline in percentage of naïve B cells out of total B cells
- Secondary and exploratory end points assessments
- Safety assessment
Joenja was studied in patients with confirmed PIK3CD or PIK3R1 variants1
Baseline demographics and disease characteristics in patients with APDS1,2
Demographics
Joenja
(n=21)
Placebo
(n=10)
Age, y (mean SD)
22.2 (10)
26.7 (13.43)
Age categories
<18 y, n (%)
(min, max)
≥18 y, n (%)
(min, max)
8 (38)
(12, 17)
13 (62)
(18, 54)
4 (40)
(15, 17)
6 (60)
(18, 48)
Sex, n (%)
Male
Female
11 (52)
10 (48)
4 (40)
6 (60)
Disease characteristics
Baseline concomitant glucocorticoids, n (%)
12
(57)
6
(60)
Baseline concomitant IgG, n (%)
14
(67)
7
(70)
Baseline antibiotic prophylaxis, n (%)
9
(42.9)
4
(40.0)
Previous sirolimus use, n (%)
4
(19)
3
(30.0)
Lymphoproliferation, n (%)
15
(71.4)
7
(70.0)
Chronic infections, n (%)
18
(85.7)
7
(70.0)
Pulmonary disease, n (%)
14
(66.7)
8
(80.0)
Asthma, n (%)
7
(33.3)
4
(40.0)
Bronchiectasis, n (%)
8
(38.1)
8
(80.0)
Cytopenias, n (%)
13
(61.9)
5
(50.0)
Gastrointestinal disease, n (%)
10
(47.6)
7
(70.0)
- More than half of patients in each treatment arm were receiving glucocorticoids, IRT, or both1,2
- Patients had nodal and/or extranodal lymphoproliferation, as measured by index nodal lesion selected by the Cheson methodology on CT or MRI, and clinical findings and manifestations compatible with APDS (eg, history of repeated oto-sino-pulmonary infections, organ dysfunction)1
- Immunosuppressive medications or PI3Kδ inhibitors (selective or nonselective) were prohibited within 6 weeks of baseline (day -1 and the visit prior to first study drug administration) and throughout the study1
- Patients who had previous or concurrent B-cell depleters (eg, rituximab) within 6 months of baseline were excluded from the study unless absolute B lymphocytes in the blood were normal. B-cell depleters were prohibited throughout the study1
APDS, activated PI3Kδ syndrome; BID, twice a day; CT, computed tomography; IgG, immunoglobulin G; IRT, immunoglobulin replacement therapy; MRI, magnetic resonance imaging; SD, standard deviation; SPD, sum of product diameters.
CO-PRIMARY END POINTS
Joenja delivers clinically proven efficacy with long-term benefits1
Log10-transformed SPD of index lesions (excluding patients with 0 lesions at baseline) at week 121*
- Improvement in lymphoproliferation as measured by a change from baseline in lymphadenopathy measured by the log10-transformed SPD of index lymph nodes1
Joenja
(n/N=18/21)†
Placebo
(n/N=8/10)†
Baseline mean (SD)
3.03 (0.42)
3.05 (0.39)
Change from baseline, LS mean (SE)
-0.27 (0.04)
-0.02 (0.05)
Difference vs placebo (95% CI)
-0.25 (-0.38,-0.12)
P=0.0006
Joenja significantly reduced lymphadenopathy, helping to restore immune balance1
- The size of swollen lymph nodes for each person in the trial was studied before and after treatment
- The results showed that lymph nodes shrank more in people taking Joenja vs those receiving placebo
At week 12, patients saw a significant reduction in lymphadenopathy‡ with Joenja vs placebo1,5
That is an ~8x greater improvement with Joenja vs placebo
Joenja significantly increased naïve B cells2
Significantly improved immunophenotype vs placebo at week 121,2
- In patients with <48% of naïve B cells at baseline,§ the adjusted mean difference between Joenja (n=8) and placebo (n=5) in the percentage of naïve B cells out of total B cells was 37.30 (95% CI: 24.06, 50.54), P=0.0002||
- The adjusted mean change from baseline (SE) for Joenja was 37.39 (5.35) and 0.09 (6.66) for placebo
Absolute percentage of naïve B cells over time2||¶
Mean naïve B-cell levels within normal range by week 4 and maintained through week 12 with Joenja
CI, confidence interval; IRT, immunoglobulin replacement therapy; LS, least squares; SE, standard error; SPD, sum of product diameters.
*
The LS mean change from baseline, difference in LS mean change from baseline between Joenja and placebo and its P value, were obtained from an ANCOVA model with treatment, glucocorticoid use and immunoglobulin replacement therapy at baseline, and baseline measurement as covariates.1
†
The analysis excluded 2 patients from each treatment group due to protocol deviations and 1 Joenja patient having complete resolution of the index lesion identified at baseline.1
‡
The LS mean change from baseline, difference in LS mean change from baseline between Joenja and placebo and its P value, were obtained from an ANCOVA model with treatment, glucocorticoid use and IRT at baseline, and baseline measurement as covariates.1
§
The analysis excluded 2 patients from each treatment group due to protocol deviations and 1 Joenja patient having complete resolution of the index lesion identified at baseline.1
||
Normal range for percentage of naïve B cells indicated by shaded bar in graph.1
¶
Cell surface markers used to distinguish naïve B cells on flow cytometry were CD19+, CD27-, and CD10-. Baseline is defined as the arithmetic mean of the baseline and day 1 values when both were available, and if either was missing, the existing value was used.1
The analysis excluded 2 patients from each treatment group due to protocol deviations, 5 Joenja patients and 3 placebo patients with ≥48% naïve B cells at baseline, 5 Joenja patients with no day 85 measurement, and 1 Joenja patient with no baseline measurement.1
SECONDARY & EXPLORATORY ENDPOINTS
Joenja significantly reduced splenomegaly2
Significant reductions in spleen size by 2D and 3D analysis compared to placebo
- The adjusted mean difference in bidimensional spleen size between Joenja (n=19) and placebo (n=9) was -13.5 cm2 (95% CI: -24.1, -2.91), P=0.0148
- The adjusted mean difference in 3D spleen volume between Joenja (n=19) and placebo (n=9) was -186 cm3 (95% CI: -297, -76.2), P=0.0020
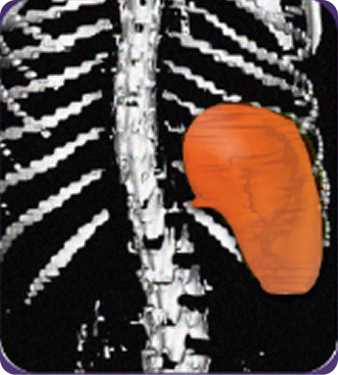
Secondary measure: Spleen volume scan results of actual patient illustrate average improvement documented for patients taking Joenja2,6
Prior to treatment
491 mL
At week 12
314 mL
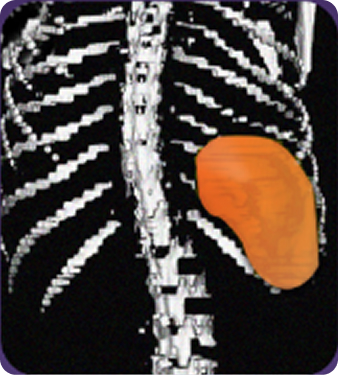
Actual images of a 29-year-old female’s response of spleen size reduction, representing the average response in the study. As individual results vary, images may not be representative of all patients.
Other secondary end points included patient-reported benefits with Joenja that were assessed using the SF-36 (Short Form 36) Survey and WPAI-CIQ (Work Productivity Activity Impairment plus Classroom Impairment Questionnaire), visual analogue scales for Physician’s Global Assessment (PGA), and Patient’s Global Assessment (PtGA), and patient narratives by investigator. Clinical relevance from these assessments was not established.7
An exploratory end point showed Joenja reduced IgM levels2
Mean serum IgM reduced6
- In the Joenja arm, IgM was elevated above normal limits in 6 patients at baseline, and by week 12 was reduced in all, with 50% returning to within normal limits
- In contrast, IgM was elevated above normal limits at baseline in 4 patients in the placebo arm, and by day 85, levels remained stable or elevated, with 0% returning to within normal limits
- Soluble biomarkers, including IgM, were prespecified exploratory end points in the protocol. Although an observational decrease in IgM was noted in some patients, no statistical significance can be made from this analysis, and no conclusions should be drawn2
Mean serum IgM reduced to within normal limits
Normal range for IgM indicated by shaded bar in graph
ANCOVA, analysis of covariance; CI, confidence interval; IgM, immunoglobulin M; IV, intravenous; PD, pharmacodynamics; SD, standard deviation.
This analysis excluded 2 patients in each treatment group. In the Joenja group, 1 patient with a complete index lesion response was excluded, and 3 patients were excluded for no non-index lesion at baseline.1
#
In the PD analysis set, the mean (SD) percentage change from baseline to week 12 in 3D spleen volume (mm3) was -26.68% (12.137) with Joenja (n=19) and -1.37% (24.238) with placebo (n=9). The ANCOVA model was used with treatment as a fixed effect and log10-transformed baseline as a covariate for index and non-index lesions. The use of both glucocorticoids and IV immunoglobulin at baseline was included as categorical (yes/no) covariates.6
PIVOTAL TRIAL SAFETY
Joenja safety results in the randomized, placebo-controlled pivotal study
- No serious adverse drug reactions were reported2
- No patients withdrew due to an adverse drug reaction2
- The most common adverse reactions (>10%) were headache, sinusitis, and dermatitis atopic1
Adverse reactions reported by ≥2 Joenja-treated patients and more frequently than placebo1
Joenja
(n=21)
n (%)
Placebo
(n=10)
n (%)
Headache
5 (24%)
2 (20%)
Sinusitis
4 (19%)
0 (0%)
Dermatitis atopic*
3 (14%)
0 (0%)
Tachycardia†
2 (10%)
0 (0%)
Diarrhea
2 (10%)
0 (0%)
Fatigue
2 (10%)
1 (10%)
Pyrexia
2 (10%)
0 (0%)
Back pain
2 (10%)
0 (0%)
Neck pain
2 (10%)
0 (0%)
Alopecia
2 (10%)
0 (0%)
*
Dermatitis atopic: including dermatitis atopic and eczema.1
†
Tachycardia: including tachycardia and sinus tachycardia.1
OLE SAFETY DATA
Additional safety results from an interim analysis1,4
At the data cutoff1,4
- Thirty-seven of 38 patients received Joenja 70 mg orally twice daily for at least 25 weeks; 66% were exposed for 96 weeks or longer
- Median duration of Joenja treatment was approximately 2 years1
- Four patients had more than 5 years of Joenja exposure
- In the open-label clinical trial, five patients (14%) experienced weight gain
Most common AEs
n
Upper respiratory tract infection
9
Headache
6
Pyrexia
6
Otitis externa
5
Weight increase
5
COVID-19 positive
5
COVID-19 negative
14
- Thirty-two of 37 patients had ≥1 AE (333 AEs reported)5,8
- 78.4% were grade 1, 48.6% were grade 2, and 27% were grade 38
- No grade 4 AEs reported8
- One grade 5 patient with significant baseline comorbidities suffered cardiac arrest resulting in death on day 879; investigator determined that the death was not related to study drug8
- No serious AEs were related to Joenja treatment8
- The AEs reported as related to study drug were weight increase (3 patients), arthralgia (1 patient), hyperglycemia (1 patient), and decreased neutrophil count (1 patient)8
Four patients had more than 5 years of Joenja exposure1
ADDITIONAL FINDINGS FROM THE OLE STUDY
Annual infection rates
Infection rates reported per each additional year of treatment with Joenja9
- Infections that developed during the study were reported as AEs. Investigators were requested to inquire about signs and symptoms of infections at each visit, in particular bacterial enterocolitis
- Patients were not provided an infection diary to document infections occurring between visits
- No change in antibiotic use was seen despite the reduction in IRT utilization
- Although safety was the primary objective of the open-label extension (OLE) study, this post hoc analysis from the OLE study was not powered to provide any statistical significance of efficacy; therefore, no conclusions should be drawn
Physician-reported IRT reductions and discontinuations
- Data on concomitant medication usage were recorded at each patient visit
- Four patients had been IRT-free for 1 to 2.5 years‡
- The median time to IRT reduction was 12.1 months, and the median time to IRT discontinuation was 11.9 months
- IRT use was captured by the investigator as concomitant medication at each study visit per protocol in this open-label study. IRT was not prespecified as an end point or analysis. This is an observation from a post hoc analysis, and no determination of statistical significance can be made and no conclusions should be drawn.
Twenty-seven Joenja patients were receiving IRT at the start of the OLE study
AT THE DATA CUTOFF
AE, adverse event; IRT, immunoglobulin replacement therapy.
*
CTCAE were used to determine AE grade. If CTCAE grading did not exist for an AE, the following definitions were used: 1, mild; 2, moderate; 3, severe; 4, life-threatening; 5, death.
†
m/N, number of infection days/number of patients in follow-up category.
‡
Baseline infections are each group’s year 1 annualized rate of infections. N values changed because patients were in the study for different lengths of time.
§
One of these patients had a subsequent one-time dose of IRT.
Data analyzed using a log-linear negative binomial model including an offset for time spent in study, an effect for time of the start of infection (in years), and presence of baseline infection as a covariate. One patient was excluded from the analysis due to a wrong year recorded for an infection.
Efficacy results
(For children ages 4-11)
→
Contact a rep
→
References: 1. Joenja (leniolisib). Prescribing information. Pharming Healthcare, Inc; 2025. 2. Rao VK, Kulm E, Šediv. A, et al. Interim analysis: Open label extension study of leniolisib for patients with APDS. J Allergy Clin Immunol. 2024;153(1):265-274.e9. doi:10.1016/j.jaci.2023.09.032 3. Rao VK, Webster S, Dalm VASH, et al.Effective “activated PI3Kδ syndrome”—targeted therapy with the PI3Kδ inhibitor leniolisib. Blood. 2017;130(21):2307-2316. doi:10.1182/blood-2017-08-80119 4. Rao VK, Kulm E, Šedivá A, et al. Interim analysis: open-label extension study of leniolisib for patients with APDS. J Allergy Clin Immunol. 2024;153(1):265-274.e9. doi:10.1016/j.jaci.2023.09.032 5. Rao VK, Webster S, Šediv. A, et al. A randomized, placebo-controlled phase 3 trial of the PI3Kδ inhibitor leniolisib for activated PI3Kδ syndrome. Blood. 2023;141(9):971-983. doi:10.1182/blood.2022018546 6. Data on file. Pharming Healthcare, Inc. 7. Rao VK, Webster S, Šediv. A, et al. A randomized, placebo-controlled phase 3 trial of the PI3Kδ inhibitor leniolisib for activated PI3Kδ syndrome. Supplemental. Blood. 2023;141(9):971-983. doi:10.1182/blood.2022018546 8. Boyle A. NIH study leads to first FDA approval for rare immunodeficiency syndrome. U.S. Medicine. Published June 19, 2024. Accessed January 9, 2025. https://www.usmedicine.com/2024-compendium-of-federal-medicine/nih-study-leads-to-first-fda-approval-for-rareimmunodeficiency-syndrome/ 9. Rao VK. Interim analysis of safety and hematological parameters of an ongoing long-term open-label extension study of investigational PI3Kδ inhibitor leniolisib for patients with activated PI3K delta syndrome (APDS). Deck presented at: The 64th American Society of Hematology (ASH) Annual Meeting and Exposition; December 10-13, 2022; New Orleans, LA.